|
Changes of state in volume-pressure diagrams This ultimately leads to the concept of pressure-volume work. Therefore, the release or absorption of work during the change of state of a gas will be discussed in more detail in this article. Thus, gases play a central role in both heat engines and heat pumps. Figure: Operating principle of a heat pump (conversion of work into heat) The gas therefore converts work into heat. From the point of view of the gas, therefore, work is done on to the gas on the one hand, while the gas in turn releases heat. If the outlet valve is closed during pumping, not only does the pressure rise very sharply, but an increase in temperature can also be observed due to the work done on the gas during compression. The principle of a heat pump can be illustrated using an air pump. the conversion of work into heat for heating purposes, on the other hand, takes place in so-called heat pumps. Figure: Operating principle of a heat engine (conversion of heat into work) For this reason, internal combustion engines are also generally referred to as heat engines. 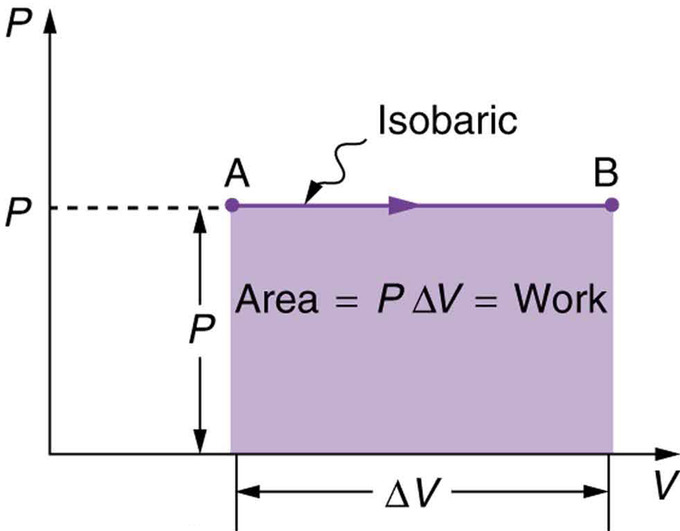
From the point of view of the gas, therefore, heat is supplied to the gas on the one hand, while on the other hand work is done by the gas. 
This leads to an increase in pressure in the cylinder, which pushes the piston down and drives the crankshaft. 
The temperature of the gaseous fuel-air mixture rises due to heat released during the combustion of the fuel. In internal combustion engines, for example, they convert heat into work. Gases play an important role in thermodynamics.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
